Nylon
 One example of nylon (courtesy of pcls.ws)
One example of nylon (courtesy of pcls.ws)
Abundance
Though not commonly found in everyday wardrobe pieces, nylon is fundamental to our jackets, synthetic wool sweaters, and many other types of waterproof clothing. Outside of our wardrobes, nylons make up our umbrellas, and increasingly, our car tires. Nearly 4 million tons of nylon were produced in 2005 alone, yet figures indicate this number is dropping 0.35% each year [1].
History
Developed by scientists at DuPont in the early 1930s, nylon was the first synthetic fiber to be commercialized in 1939 [2]. First used in WWII, nylon quickly became popular in the production of parachutes and tires for its durability and long-lifespan [3]. By 1970, approximately 40% of all synthetic fibers were nylon [4].
Materials
Nylon is a polyamide fiber (literally translated, a fiber comprised of many amides), derived from a diamine and a dicarboxylic acid. There are a wide variety of diamines and dicarboxylic acids, creating a plethora of unique nylon fibers. However, the two most common version are nylon 6,6 (polyhexamethylene adiamide) and nylon 6 (Polycaprolactam, a cyclic nylon intermediate).
Manufacturing
Though nylon may be made into a wide variety of fibers for various things like ropes, umbrellas, tires, or clothes, the initial chemical process is all the same.
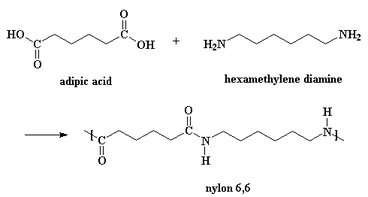
As mentioned above, there are a wide variety of diamines and dicarboxylic acids. However, a common reaction used to synthesize nylon uses hexamethylene diamine monomers, and adipic acid (see diagram on left) [3].
1) Cyclohexane is oxidized by air, then nitric acid, to produce adipic acid. Adiponitrile is hydrogenated to form hexamethylene diamine [5].
2) Adipic acid and hexamethylen diamine are then reacted together to form nylon salt.
3) 18 atm of pressure, and a heat of 280 C is applied to the nylon salt. As the water evaporates, long chains of nylon salt form nylon.
4) At this stage, additives and pigmentation can be added to change the properties of nylon depending on what its ultimate use is determined to be.
4) Once molten, the nylon polymers are forced through fine sieves and is cooled in water
5) Once hardened by the cooling, nylon is chopped into granules 3-4 mm long
5) The granules leave the production site. At their new factory, they are remelted and forced through the holes of a fine metal sieve [3].
6) The resulting fibers are forced into a spinneret, separating the fibers into thin filaments and exposing it to the air, causing immediate hardening.
7) Once hardened, the filaments are wound onto bobbins and drawn. This refers to a process in which filaments are then rewound onto a smaller spool for the purpose of aligning the molecules in parallel structure.
8) The filaments are now ready to be fused or woven together to form thicker threads.
Nylon filaments may be woven in with other materials, such as cotton, making the resulting material strong and water resistant from the nylon, but soft to the touch from the cotton. Filaments can also be woven into different patterns to reinforce strength or improve the visual aesthetics of the material.
Environmental Impact
Though nylon is durable and requires minimal maintenance, it has many drawbacks on the environment. Like other synthetic fibers, it is not biodegradable and is composed of compounds made from petroleum [6]. However, what makes nylon an even bigger environmental culprit is the fact that producing adipic acid (a vital step in nylon synthesis) releases noxious nitrous oxide, a greenhouse gas [7]. Though factories have reduced nitrous oxide emissions 61 percent between 1990 and 2006, the production of adipic acid accounts for 5-8 percent of human emissions of nitrous oxide.
Another negative aspect of nylon is that it cannot be melted down and recycled. Unless completely broken down into its original chemicals again (which virtually defeats the purpose of recycling with the resources and energy required to do this), nylon can only be repurposed, not rewoven and completely reused [4].
What You Can Do
Don't by regular spandex if you can help it! However, stay tuned for "sustainable nylon". A non-naturally occurring microbial organism has been patented by a start-up company in San Diego called Genomatica, which produces adipic acid among other compounds [8]. This would eliminate much of the need for petroleum products, as well as stop production of nitrous oxide in nylon synthesis. Unfortunately, fixing one of the most harmful steps of nylon will not make nylon a completely eco-friendly fabric, for it still consumes an incredible amount of energy to melt and remelt, spin, and re-spin, and just as importantly, it would still not be biodegradable. So, while nylon may be getting greener in the near future, it is still a material to be avoided.
Though not commonly found in everyday wardrobe pieces, nylon is fundamental to our jackets, synthetic wool sweaters, and many other types of waterproof clothing. Outside of our wardrobes, nylons make up our umbrellas, and increasingly, our car tires. Nearly 4 million tons of nylon were produced in 2005 alone, yet figures indicate this number is dropping 0.35% each year [1].
History
Developed by scientists at DuPont in the early 1930s, nylon was the first synthetic fiber to be commercialized in 1939 [2]. First used in WWII, nylon quickly became popular in the production of parachutes and tires for its durability and long-lifespan [3]. By 1970, approximately 40% of all synthetic fibers were nylon [4].
Materials
Nylon is a polyamide fiber (literally translated, a fiber comprised of many amides), derived from a diamine and a dicarboxylic acid. There are a wide variety of diamines and dicarboxylic acids, creating a plethora of unique nylon fibers. However, the two most common version are nylon 6,6 (polyhexamethylene adiamide) and nylon 6 (Polycaprolactam, a cyclic nylon intermediate).
Manufacturing
Though nylon may be made into a wide variety of fibers for various things like ropes, umbrellas, tires, or clothes, the initial chemical process is all the same.
As mentioned above, there are a wide variety of diamines and dicarboxylic acids. However, a common reaction used to synthesize nylon uses hexamethylene diamine monomers, and adipic acid (see diagram on left) [3].
1) Cyclohexane is oxidized by air, then nitric acid, to produce adipic acid. Adiponitrile is hydrogenated to form hexamethylene diamine [5].
2) Adipic acid and hexamethylen diamine are then reacted together to form nylon salt.
3) 18 atm of pressure, and a heat of 280 C is applied to the nylon salt. As the water evaporates, long chains of nylon salt form nylon.
4) At this stage, additives and pigmentation can be added to change the properties of nylon depending on what its ultimate use is determined to be.
4) Once molten, the nylon polymers are forced through fine sieves and is cooled in water
5) Once hardened by the cooling, nylon is chopped into granules 3-4 mm long
5) The granules leave the production site. At their new factory, they are remelted and forced through the holes of a fine metal sieve [3].
6) The resulting fibers are forced into a spinneret, separating the fibers into thin filaments and exposing it to the air, causing immediate hardening.
7) Once hardened, the filaments are wound onto bobbins and drawn. This refers to a process in which filaments are then rewound onto a smaller spool for the purpose of aligning the molecules in parallel structure.
8) The filaments are now ready to be fused or woven together to form thicker threads.
Nylon filaments may be woven in with other materials, such as cotton, making the resulting material strong and water resistant from the nylon, but soft to the touch from the cotton. Filaments can also be woven into different patterns to reinforce strength or improve the visual aesthetics of the material.
Environmental Impact
Though nylon is durable and requires minimal maintenance, it has many drawbacks on the environment. Like other synthetic fibers, it is not biodegradable and is composed of compounds made from petroleum [6]. However, what makes nylon an even bigger environmental culprit is the fact that producing adipic acid (a vital step in nylon synthesis) releases noxious nitrous oxide, a greenhouse gas [7]. Though factories have reduced nitrous oxide emissions 61 percent between 1990 and 2006, the production of adipic acid accounts for 5-8 percent of human emissions of nitrous oxide.
Another negative aspect of nylon is that it cannot be melted down and recycled. Unless completely broken down into its original chemicals again (which virtually defeats the purpose of recycling with the resources and energy required to do this), nylon can only be repurposed, not rewoven and completely reused [4].
What You Can Do
Don't by regular spandex if you can help it! However, stay tuned for "sustainable nylon". A non-naturally occurring microbial organism has been patented by a start-up company in San Diego called Genomatica, which produces adipic acid among other compounds [8]. This would eliminate much of the need for petroleum products, as well as stop production of nitrous oxide in nylon synthesis. Unfortunately, fixing one of the most harmful steps of nylon will not make nylon a completely eco-friendly fabric, for it still consumes an incredible amount of energy to melt and remelt, spin, and re-spin, and just as importantly, it would still not be biodegradable. So, while nylon may be getting greener in the near future, it is still a material to be avoided.
