Polyester
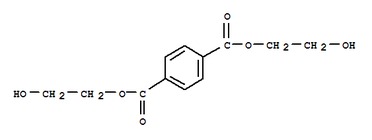 bis-(2-hydroxyethyl)terephthalate, the building block of polyester
bis-(2-hydroxyethyl)terephthalate, the building block of polyester
Abundance
Polyester one of the most abundant synthetic fabrics on the market, comprising over 70% of all synthetic fibers [1] and over 45% of all fibers as of 2004 [2], though these figures have grown quickly over the past decade and are undoubtably much larger now.
History
Known for being wrinkle resistant, durable, easy to care for, and with the ability to take on many different weights and textures, polyester quickly rose to popularity among consumers. Manufacturers like polyester as well, for it is relatively simple to produce. Interestingly, PET (polyethylene terephthalate, also known as polyester) was patented in 1941 by British chemists John Whinfield and James Dickson after refining work done years before by Wallace Carothers, the inventor of nylon [3]. While nylon took the spotlight in the 1930's and 40's, polyester burst onto the American stage in 1951, hailed as a wonder fabric that needed no ironing [4].
Materials
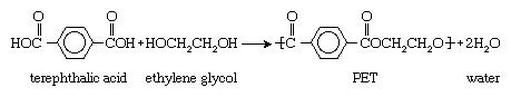
Though everyone knowns polyester is a synthetic material, hardly anyone could tell you how or what it is made of. Polyester literally means "many esters". Esters are a chemical group characterized by a carbon double bonded to an oxygen, while bonded to another oxygen attached to any alkyl or aryl group (on diagram at left, see the two carbons closest to the ring, bonded to two oxygens each). With polyester fibers, the "building block" that attaches over and over to itself to form a polyester is called bis-(2-hydroxyethyl)terephthalate. This intimidating chemical is made rather simply from two chemicals called dimethyl terephthalate (an ester) and ethylene glycol (an alcohol). Unfortunately, these chemicals are petroleum-based and have many consequences on the environment, which will be discussed later.
Manufacturing
Now that you know polyester is made from dimethyl terephthalate and ethylene glycol, you may be wondering how they're made into the clothes we wear. There are a few different ways to produce polyester depending on what the final product will be, but the two most commonly used are filament and staple form. Filament form is when each thread is continuous and uniform in length, forming smooth-surfaced fabrics. Staple form is when the filaments are cut short, making them easier to blend with other materials and create more sophisticated fabrics with a variety of different properties.
Polyester one of the most abundant synthetic fabrics on the market, comprising over 70% of all synthetic fibers [1] and over 45% of all fibers as of 2004 [2], though these figures have grown quickly over the past decade and are undoubtably much larger now.
History
Known for being wrinkle resistant, durable, easy to care for, and with the ability to take on many different weights and textures, polyester quickly rose to popularity among consumers. Manufacturers like polyester as well, for it is relatively simple to produce. Interestingly, PET (polyethylene terephthalate, also known as polyester) was patented in 1941 by British chemists John Whinfield and James Dickson after refining work done years before by Wallace Carothers, the inventor of nylon [3]. While nylon took the spotlight in the 1930's and 40's, polyester burst onto the American stage in 1951, hailed as a wonder fabric that needed no ironing [4].
Materials
Though everyone knowns polyester is a synthetic material, hardly anyone could tell you how or what it is made of. Polyester literally means "many esters". Esters are a chemical group characterized by a carbon double bonded to an oxygen, while bonded to another oxygen attached to any alkyl or aryl group (on diagram at left, see the two carbons closest to the ring, bonded to two oxygens each). With polyester fibers, the "building block" that attaches over and over to itself to form a polyester is called bis-(2-hydroxyethyl)terephthalate. This intimidating chemical is made rather simply from two chemicals called dimethyl terephthalate (an ester) and ethylene glycol (an alcohol). Unfortunately, these chemicals are petroleum-based and have many consequences on the environment, which will be discussed later.
Manufacturing
Now that you know polyester is made from dimethyl terephthalate and ethylene glycol, you may be wondering how they're made into the clothes we wear. There are a few different ways to produce polyester depending on what the final product will be, but the two most commonly used are filament and staple form. Filament form is when each thread is continuous and uniform in length, forming smooth-surfaced fabrics. Staple form is when the filaments are cut short, making them easier to blend with other materials and create more sophisticated fabrics with a variety of different properties.
Regardless of the form, the basic process still begins the same:
1) The dimethyl terephthalate and ethylene glycol are reacted with the help of an antimony catalyst, either antimony trioxide (Sb2O3) or antimony triacetate (Sb(CH3COO)3). This is done at 302-410°F (150-210°C) and results in dimethyl terephthalate.
2) Then at 472°F (280°C), the dimethyl terephthalate produced in the first step is reacted with more ethylene glycol to form the bis-(2-hydroxyethyl)terephthalate, the building block of polyester.
3) The bis-(2-hydroxyethyl)terephthalate reacts with itself many times, forming clear, molten polyester (also known as polyethylene terephthalate, or PET) that is then forced through a sieve into long ribbons.
4) The ribbons are cooled until they are brittle, then cut up into little chips and dried to prevent any differences in consistency.
5) The chips are melted into a molten solution at 500-518°F (260-270°C).
6) The molten, syrupy solution is forced through a tiny sieve, with the size of the holes dictating the size of the resulting yarn and ultimate threads. At this spinning stage, a variety of other chemicals may be added to the solution, making the resulting polyester flame retardant, antistatic, or easier to dye.
7) After emerging yarn is brought together to form threads, it is soft, can easily stretch up to five times its original length, and is not strong. To remedy this, the threads are heated and stretched to align the polymers (PET) into parallel formation.
8) The fiber is now torn apart, and the resulting fluff is the raw material for making useable polyester.
9) This cotton/wool looking fluff is then sent to a different factory, where it is scraped into a rough cloth and carded so that all bonded fibers are in the same direction so that it is strong and durable.
10) The rough cloth is then spun and collected on bobbins, essentially enormous spools of thread, finally ready to be woven into a fabric of polyester.
11) To make the material softer and smoother, another machine takes the fabric and makes tiny loops with the thread in the fabric. Another machine then catches and breaks these loops, making it softer to the touch, and it is now ready to be sent to clothing factories and finally made into clothes.
Read more about the process: http://www.madehow.com/Volume-2/Polyester.html#ixzz2vJ08IPdbRead more about the chemistry: http://www.pslc.ws/mactest/petsyn.htm
If you think that all this shipping, heating, cooling, reheating, etc. seems like a lot of energy, you'd be right. But there is another step that also takes a lot of energy: dyeing polyester. Polyester is difficult to dye because of its relatively high hydrophobicity (meaning it does not accept water very easily) and crystallinity (meaning its polymers are highly ordered, making it even harder for dyes to penetrate). As such, it has to be heated again, usually to about 266°F (130ºC) and treated harshly for a relatively long period of time [5]. However, just a few years ago, the MARA University of Technology in Malaysia developed a dye using the shells of shrimp and other crustaceans that lowers the temperature polyester must be dyed at [5]. While this may be a way to lower factory energy costs, there is doubt that using dye from shells is any more eco-friendly.
1) The dimethyl terephthalate and ethylene glycol are reacted with the help of an antimony catalyst, either antimony trioxide (Sb2O3) or antimony triacetate (Sb(CH3COO)3). This is done at 302-410°F (150-210°C) and results in dimethyl terephthalate.
2) Then at 472°F (280°C), the dimethyl terephthalate produced in the first step is reacted with more ethylene glycol to form the bis-(2-hydroxyethyl)terephthalate, the building block of polyester.
3) The bis-(2-hydroxyethyl)terephthalate reacts with itself many times, forming clear, molten polyester (also known as polyethylene terephthalate, or PET) that is then forced through a sieve into long ribbons.
4) The ribbons are cooled until they are brittle, then cut up into little chips and dried to prevent any differences in consistency.
5) The chips are melted into a molten solution at 500-518°F (260-270°C).
6) The molten, syrupy solution is forced through a tiny sieve, with the size of the holes dictating the size of the resulting yarn and ultimate threads. At this spinning stage, a variety of other chemicals may be added to the solution, making the resulting polyester flame retardant, antistatic, or easier to dye.
7) After emerging yarn is brought together to form threads, it is soft, can easily stretch up to five times its original length, and is not strong. To remedy this, the threads are heated and stretched to align the polymers (PET) into parallel formation.
8) The fiber is now torn apart, and the resulting fluff is the raw material for making useable polyester.
9) This cotton/wool looking fluff is then sent to a different factory, where it is scraped into a rough cloth and carded so that all bonded fibers are in the same direction so that it is strong and durable.
10) The rough cloth is then spun and collected on bobbins, essentially enormous spools of thread, finally ready to be woven into a fabric of polyester.
11) To make the material softer and smoother, another machine takes the fabric and makes tiny loops with the thread in the fabric. Another machine then catches and breaks these loops, making it softer to the touch, and it is now ready to be sent to clothing factories and finally made into clothes.
Read more about the process: http://www.madehow.com/Volume-2/Polyester.html#ixzz2vJ08IPdbRead more about the chemistry: http://www.pslc.ws/mactest/petsyn.htm
If you think that all this shipping, heating, cooling, reheating, etc. seems like a lot of energy, you'd be right. But there is another step that also takes a lot of energy: dyeing polyester. Polyester is difficult to dye because of its relatively high hydrophobicity (meaning it does not accept water very easily) and crystallinity (meaning its polymers are highly ordered, making it even harder for dyes to penetrate). As such, it has to be heated again, usually to about 266°F (130ºC) and treated harshly for a relatively long period of time [5]. However, just a few years ago, the MARA University of Technology in Malaysia developed a dye using the shells of shrimp and other crustaceans that lowers the temperature polyester must be dyed at [5]. While this may be a way to lower factory energy costs, there is doubt that using dye from shells is any more eco-friendly.
 Notice the fabrics among the rubble (courtesy of Jennifer Savage)
Notice the fabrics among the rubble (courtesy of Jennifer Savage)
Environmental Impact
Polyester is an all around harmful material. Because it is petroleum based, it not only encourages the use of a nonrenewable resource, but it is also non-biodegradable without the help of bacteria of the genus Nocardia, and esterase (an enzyme that breaks down esters) [6]. This is not a viable option for the biodegradation of all polyester produced, and so polyester continues to flow into our land fills at astonishing rates and hardly degrades at all.
There is one redeeming quality though: polyester can be broken down into dimethyl terephthalate again, and new garments can be made with no "second-hand" look. Unfortunately, that means that to be an eco-friendly fabric it would have to be recycled 100% of the time, with no more production of new polyester. This is unlikely to happen, to say the least, yet many do advocate for recycling and progress is being made (see the recycled polyester page for more information).
What You Can Do
Despite the progress being made in recycling and awareness about the harmful ecological effects of polyester, our problems would not be solved if no one bought polyester. The truth is that the world could not keep up with demand for clothes if we all switched to organic cotton or some other organic fiber. Larger changes must be made to consumer habits for a sustainable practice to work. For example, consumers must revert back to the idea of investing in their clothes, repairing them when they are damaged instead of throwing them out, and only buying what they need. That being said, try to avoid polyester in the meantime.
